|
3/4/2023 0 Comments Quantum numbers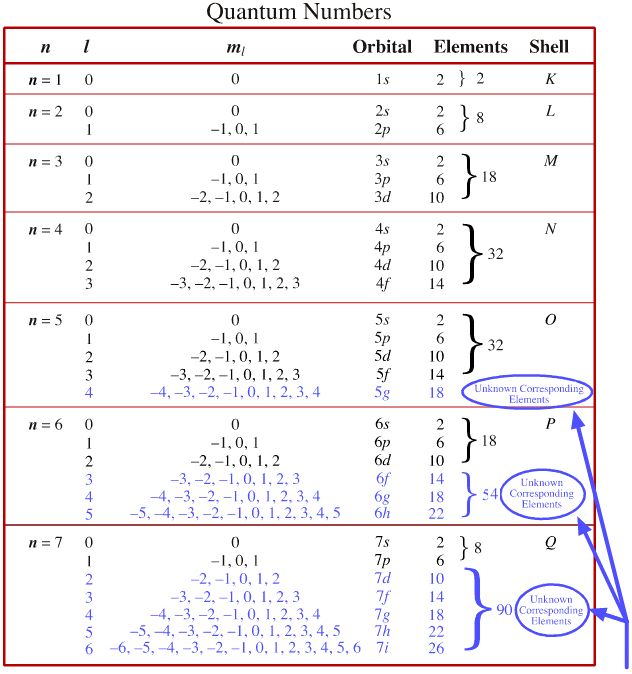
Where □ is the principal quantum number. The subsidiary quantum number can have any integer value ranging from 0 to □ − 1. The subsidiary quantum number ( □ ) describes the shape of an atomic orbital, and it is describedīy the expression □ = 0, 1, 2, 3, …, □ − 1, where □ is the principal quantum number. The 1s and 2s orbitals both have aĭefinition: Subsidiary Quantum Number (□) Subsidiary quantum number of 0 have a spherical shape and are termed s-type subshells. Subsidiary quantum number ( □ ) determines the shape of an atomic orbital. The second quantum number can be referred to as the subsidiary, azimuthal, or orbital angular momentum quantum number. We can use these statements to determine that option C is the correct answer for this question. The formula for calculating the number of orbitals can be expressedĪs □ , and the formula for calculating the total number of electrons can be expressed asĢ □ . The total number of electrons in any one energy level. Squared to determine the number of orbitals in any one energy level, and it is squared and multiplied by two to determine 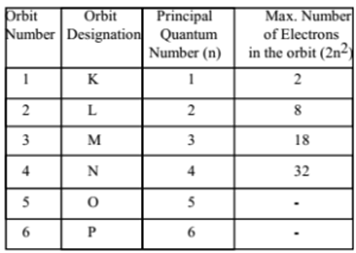
The principal quantum number ( □ ) determines the size of all the atomic orbitals, and it canīe used to determine the total number of orbitals and electrons in any one energy level. What is the relationship between the principal quantum number, □, and the total number of orbitals? The effective radius of an atomic orbital relative to the central section of an atomic nucleus.Įxample 1: Calculating the Number of Atomic Orbitals from the Principal Quantum Number The 3s atomic orbital is wider than the 2sĪtomic orbital and the 2s atomic orbital is wider than the 1s atomic orbital. Quantum numbers of □ = 2 and □ = 1 respectively. The 3s atomic orbital has a principal quantum number of □ = 3 and the 2s and 1s atomic orbitals have principal Number is always a positive integer, and it can be stated that The principal quantum number ( □ ) determines the size of an atomic orbital. Has a different set of four quantum numbers. Highest-energy electron in a potassium atom has one set of four quantum numbers, and the highest-energy electron in a cesium atom The Pauli exclusion principle states that no two electrons in any one atom can have the same set of four quantum numbers. 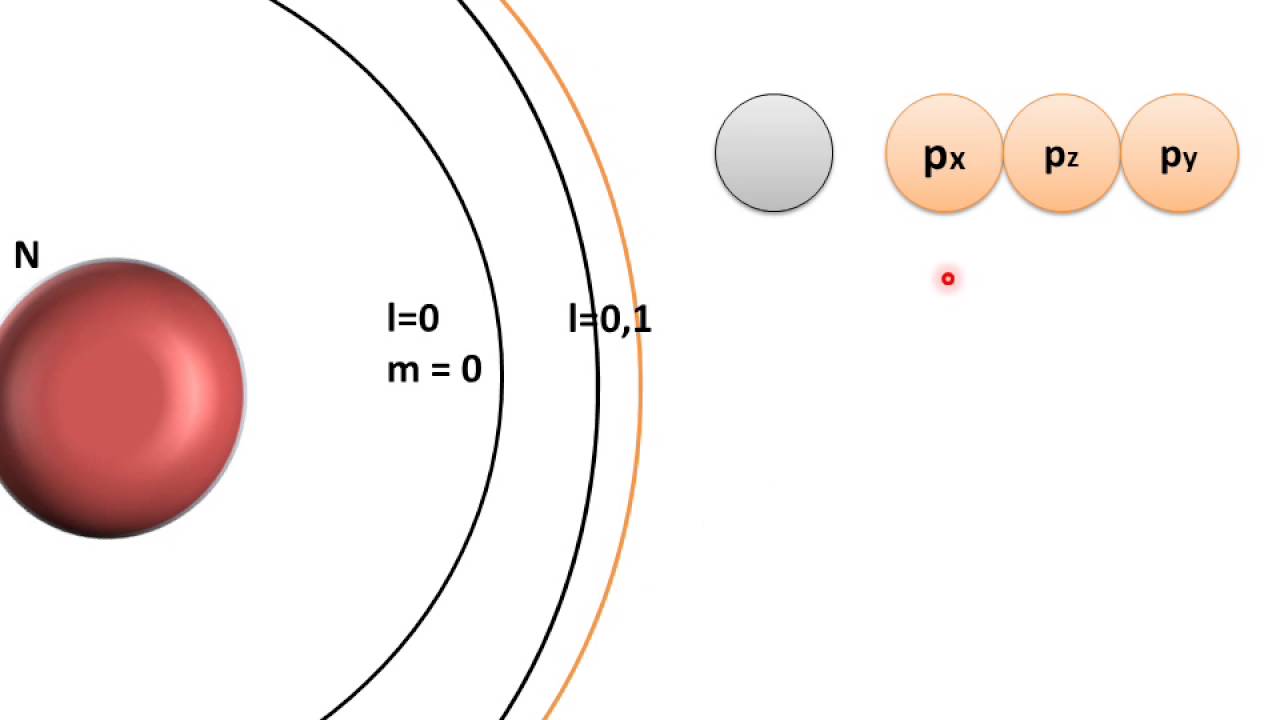
The four quantum numbers also explain why elements should be grouped into periodic table blocksĪnd why so many elements have similar chemical properties. ( □, □, □ , and □ ), and they determine how electrons In this explainer, we will learn how to use quantum numbers to describe an electron within an atom.Īn electron within an atom can be completely described with values that are known as quantum numbers.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
